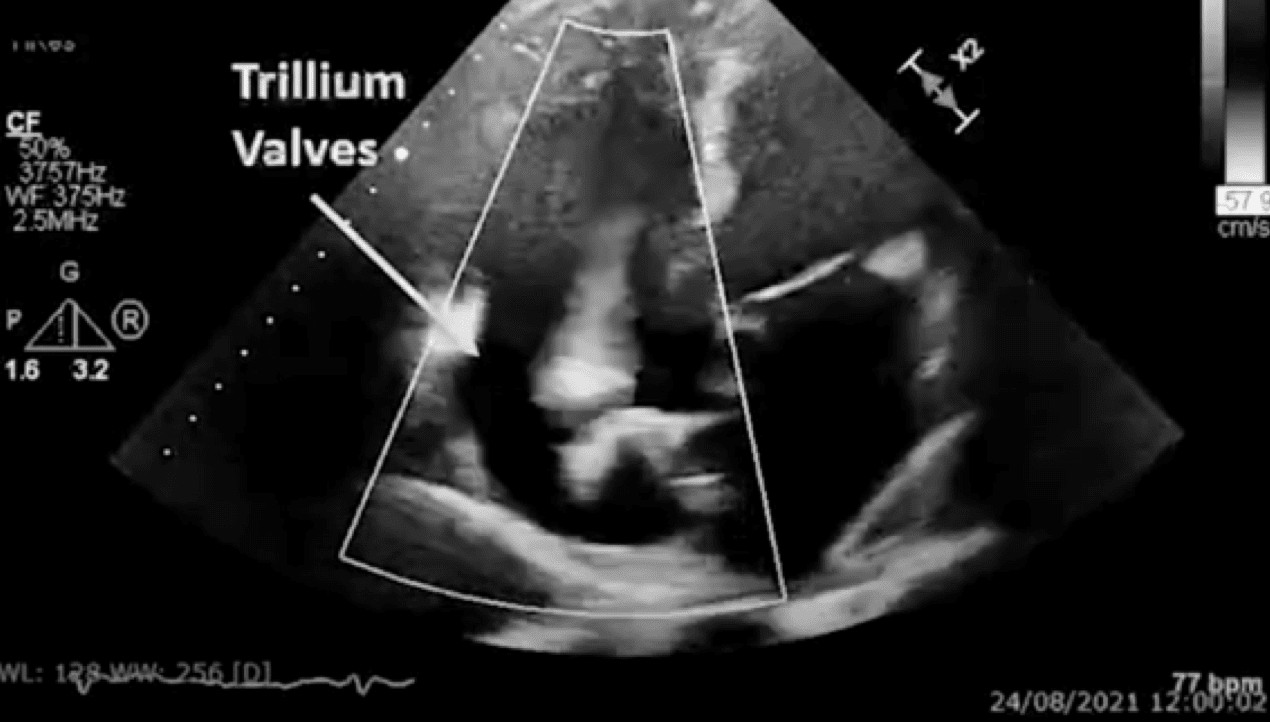
OngoingTrillium™ Early Feasibility Study (EFS)
in the treatment of severe or greater tricuspid regurgitation (TR).
Inclusion Criteria:
- Patient has clinically significant TR graded as severe or greater
- Peak central venous pressure of ≥ 15mmHg
- Patient has NYHA functional classification of III or IV
- Patient is not eligible for standard-of-care surgical or interventional therapy or has refused standard-of-care surgical and interventional therapy or has received standard-of-care TR therapy and remains symptomatic
Seattle, WA, USA
Thousand Oaks, CA, USA
Rochester, MN, USA
Detroit, MI, USA
Cincinnati, OH, USA
Nashville, TN, USA
Wynnewood, PA, USA
New York, NY, USA
Stony Brook, NY, USA
Mainz, Germany
Leipzig, Germany
Berlin, Germany
Exclusion Criteria:
Patients will be excluded from participation if ANY of the
following criteria apply:
- Severe RV dysfunction defined by TAPSE, RVEF, or RVFAC
- Non-suitable anatomy according to CT scan
- Systolic Pulmonary Artery Pressure > 65mmHg
- Moderate or more mitral valve stenosis
- Greater than moderate mitral valve regurgitation or aortic valve stenosis/regurgitation
- Moderate mitral valve regurgitation combined with moderate aortic valve stenosis/regurgitation
- Kidney dysfunction with estimated Glomerular Filtration Rate (eGFR) < 35 ml/min/1.73 m2 within 60 days prior to the index procedure or patient is on chronic dialysis
- Liver cirrhosis or moderate or severe liver disease (Child-Turcotte-Pugh class B or C, or a score of 7 or higher)
- Thrombocytopenia (Platelet count< 100,000/mm3) or thrombocytosis (Platelet count > 750,000/mm3) within 14 days of the index procedure
- In the opinion of the Investigator or the study eligibility committee, the patient’s life expectancy < 12 months
Completed enrollmentTrillium™ First-in-Human (FIH) Study
Lead PI: Dr. Philipp Lurz
Dr. Jozef Bartunek, Dr. Marc Vanderheyden
OLVZ, Aalst, Belgium
Dr. Stefan Verheye
ZNA, Antwerp, Belgium
Dr. Volker Rudolph
Heart & Diabetes Center NRW, Bad Oyenhausen, Germany
Dr. Philipp Lurz
Leipzig Heart Center, Leipzig, Germany
Dr. Mohammad Sherif
Herzzentrum der Charité (DHZC), Berlin, Germany
Dr. Rodrigo Estevez-Loureiro
Hospital Alvaro Cunqueiro, Hospital Universitario de Vigo, Spain
Dr. Ignacio Cruz Gonzales
Hospital Universitario de Salamanca, Spain
Dr. Omar Abdul-Jawad Altisent
Hospital Clinic de Barcelona, Barcelona, Spain
Dr. Ran Kornowski
Rabin Medical Center, Petah Tiqva, Israel
Saving Lives While Gaining Clinical Expertise
Our clinical experience extends beyond clinical trials, with compassionate procedures demonstrating our ability to provide quality care even to patients currently deemed untreatable.
With many successful procedures in Belgium, Spain, Germany, Georgia, and Canada, we have improved patients’ lives while showcasing the capabilities of our entire portfolio: Trillium™, Koala™, and Unica™.