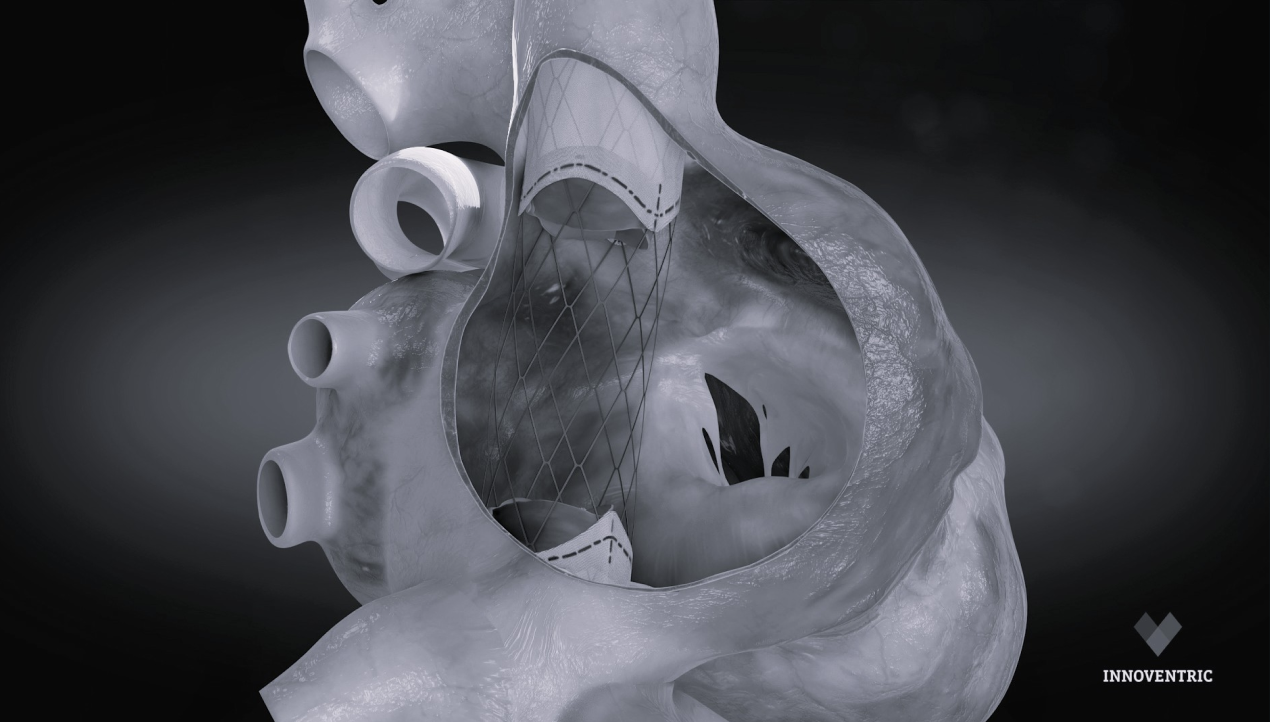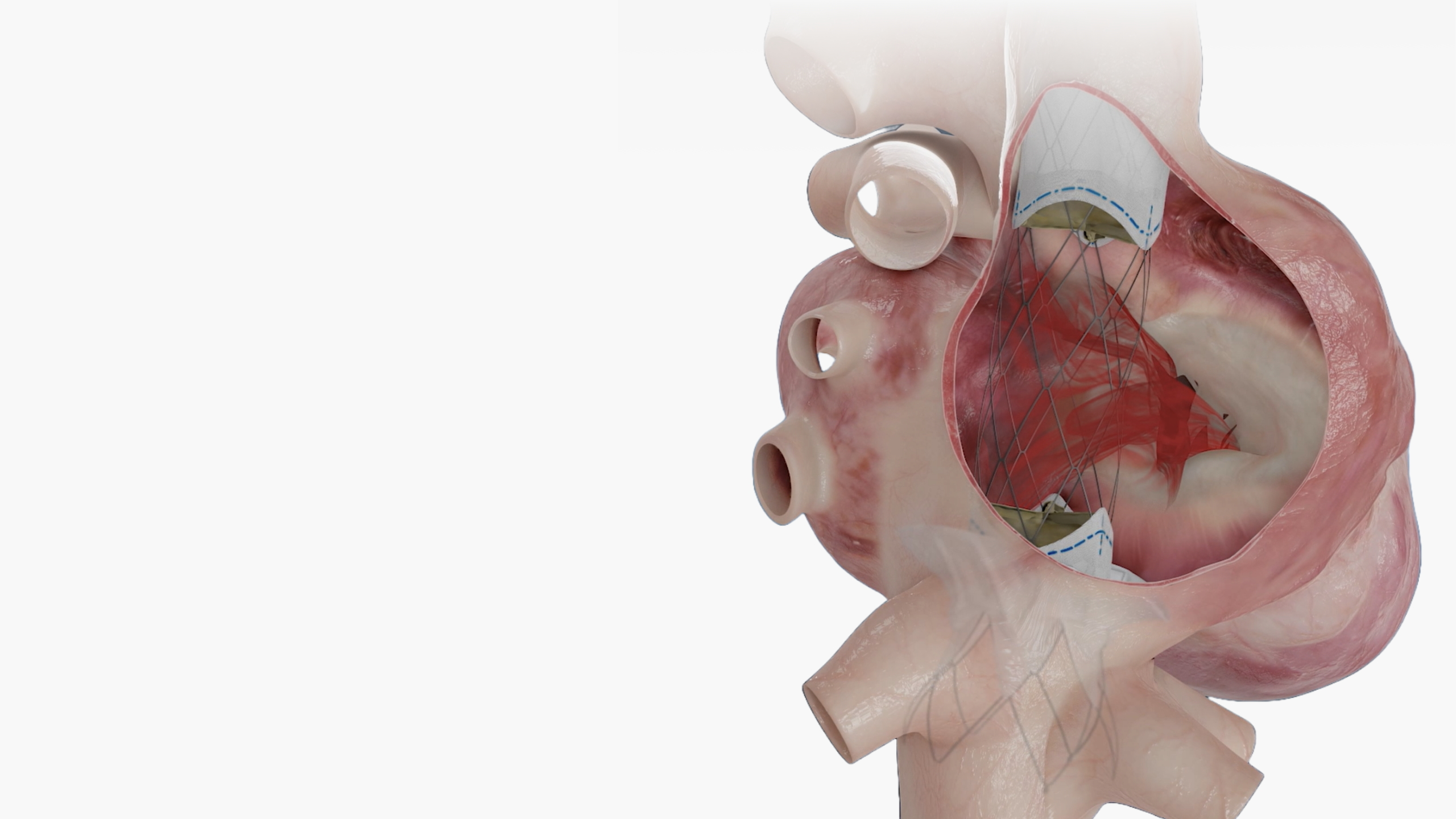
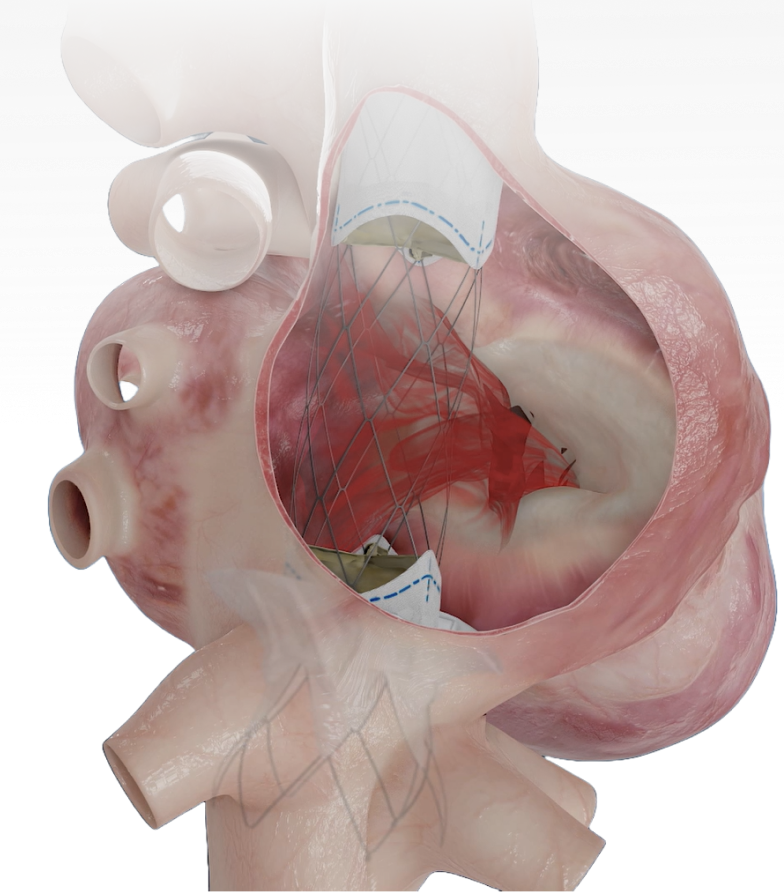


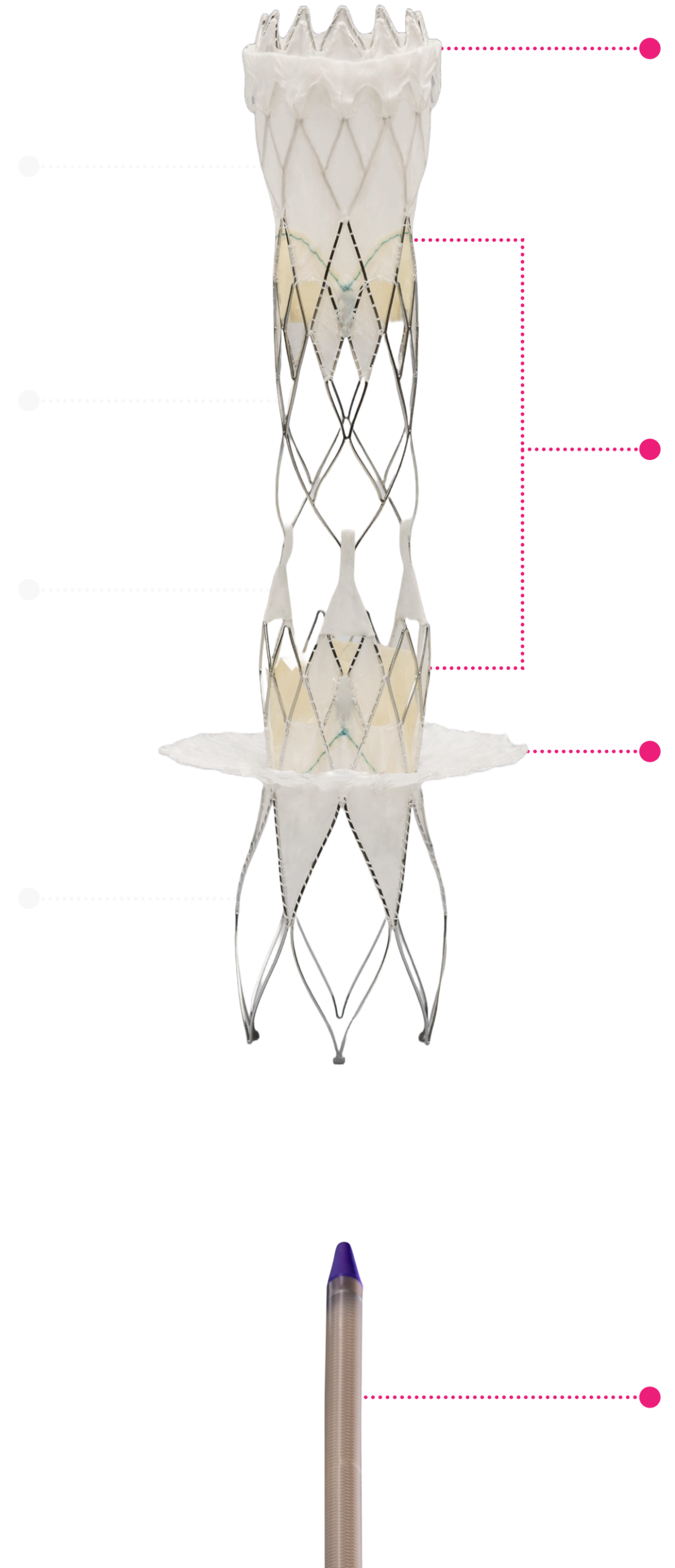
Technical Excellence for Comprehensive Care
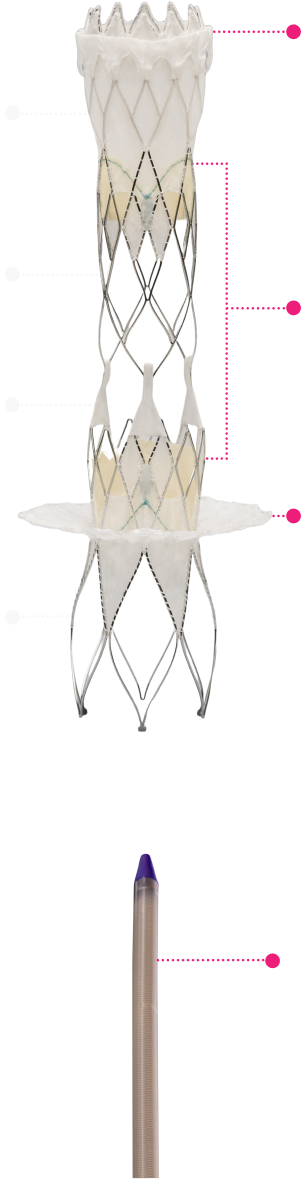
Migration Resistant
apposing anatomies (SVC & IVC),
provides reliable anchoring and full
migration resistance.
Mechanically Durable
resistance. Durable for decades of
cardiac function.
Flexible Hinge
various challenging anatomies
Broad Anatomical Reach
spectrum of anatomical sizes
SVC Skirt
Two valves
IVC Skirt
Low Profile Delivery System
Recruiting Unica™ Clinical Experience
Vast compassionate use experience
The Unica™ demonstrates unique capabilities in treating high-risk patients who were previously found ineligible for other therapies.
The Unica’s large compassionate use experience, shows compelling clinical results.
Compassionate use of the Unica™ device is available in Canada, Europe and The Middle East.
Dr. Neil Fam
ST. Michael’s Hospital, Toronto, Canada
Dr. Marino Labinaz
University of Ottawa Heart Institute, Ottawa, Canada
Dr. Rodrigo Estevez Loureiro
Ospedale Universitario Álvaro Cunqueiro,
Vigo, Spain
Dr. Didier Tchétché
Clinique Pasteur, Toulouse, France
Dr. Omar Abdul-Jawad Altisent
Hospital Clinic de Barcelona, Barcelona, Spain
Dr. Marco De Carlo
Azienda Ospedaliero Universitaria Pisana,
Pisa, Italy
Dr. Augustin Coisne
Pharmacie Centrale Secteur Essais Cliniques
CHRU de Lille, Lille, France
Dr. Stefan Verheye
ZAS Middelheim, Antwerp, Belgium
Dr. Jozef Bartunek
AZORG Hospital Aalst, Aalst, Belgium
Dr. Philipp Lurz
Universitätsmedizin Mainz, Mainz, Germany
Dr. Leor Perl
Rabin Medical Center, Petah Tikva, Israel
Ongoing FIH trial
The Unica™ is currently available at selected geographies through a First-In-Human (FIH) study, and enrollment completion is anticipated by the end of 2026. The results from the First-In-Human (FIH) study combined with the many compassionate use case performed, will serve as the basis for the approval of a global study in the near future.